Quality as a Competitive Advantage: Rebecca Crane on Building Neurotech That Works
When people think about what makes a medical device company succeed, they usually go straight to the technology, the fundraise, or the regulatory path. Rebecca Crane thinks the real answer often starts earlier than that.
As CEO and Managing Partner of Crane Medtech Partners, Rebecca has spent more than a decade working across quality and business operations in medtech, with experience at companies including Advanced Bionics, Medtronic, SetPoint Medical, and iota Biosciences. She has worked inside big, established organizations and early-stage startups, and has seen firsthand how differently teams think about Quality depending on their stage, pace, and pressure.
What comes through quickly in speaking with her is that she does not see Quality as paperwork or a box-ticking function — she sees it as something much more practical and much more important. In fast-moving neurotech, where timelines are tight and the technology is often pushing into new territory, Quality can either become a burden or a real advantage. Rebecca is firmly in the second camp.
In this conversation, she talks about what Quality actually looks like inside startups, why so many founders leave it too late, and how the role of Quality is changing as neurotech becomes more software-heavy, more complex, and more shaped by sensing, AI, and closed-loop systems.

Rebecca Crane is CEO and Managing Partner of Crane Medtech Partners.
What originally drew you into medtech — and eventually neurotech — and how did those early experiences shape your view of product development and patient impact?
My path into medtech started at Caltech, where I studied Computation and Neural Systems. From there, I joined Advanced Bionics, and what really drew me in was the bigger purpose behind the work. I still remember meeting patients with cochlear implants and hearing them talk about what it meant to hear for the first time. Those moments stay with you.
Later, at SetPoint Medical, we were developing a bioelectronic system designed to modulate the body’s inflammatory reflex through the nervous system. Hearing about patients who had been so limited in their daily lives suddenly being able to walk or bike again was incredible. At iota Biosciences, we were working across multiple patient indications at once, which brought another layer of complexity and excitement.
At the same time, product development is hard. It takes a long time, and there is usually a lot more grind than glamour. Those experiences taught me that it is not enough to have a novel idea or exciting technology. You really have to understand the patient journey, and you need the conviction to keep iterating until you have something that actually works in the real world.
You have worked at Advanced Bionics, iota Biosciences, and SetPoint Medical. What lessons from those environments most influenced your approach to Quality — particularly in fast-moving innovations like implants, neuromodulation, and bioelectronic devices?
Each company taught me something different.
At Advanced Bionics and Medtronic, I learned what Quality looks like in mature organizations. Those are environments with strong systems, a lot of rigor, and a very clear understanding that one issue can have huge consequences. That was incredibly valuable because it showed me why structure and discipline matter when you are building things that directly affect patients.
“ Quality should support innovation, not get in the way of it. ”
At SetPoint Medical, and especially at iota Biosciences, I saw something different. Startups are moving fast, resources are tighter, and teams are often building something genuinely new. In that world, Quality cannot just be a slow, bureaucratic process layered on top. It has to be useful. It has to be built in early, but in a way that still fits the pace of the company.
That is probably the biggest lesson I took from those environments. Quality should support innovation, not get in the way of it. It should help teams think clearly about risk, solve problems quickly, and make better decisions. Sometimes that means digging into why a device failed, why a weld cracked, or what to do when a key supplier changes something unexpectedly. The best Quality work happens when it is right there with engineering — not operating separately from it.
In your view, what is the biggest misconception about Quality in medtech and neurotech?
The biggest misconception is that Quality is mostly about documentation and compliance. A lot of people think it is just about checking boxes, preparing for audits, and keeping records in order.
That is part of it, obviously, but it is not the part I find most interesting, and it is definitely not the part that matters most in a startup.
In practice, especially in early-stage companies, Quality is often much more about helping the team figure out what needs to happen and how to do it well. It is about understanding which requirements matter now, which risks need to be addressed early, and how to build in a way that does not create bigger problems later. A lot of the time, it is basically problem-solving alongside the engineering team.
That is the version of Quality I care most about. It is not there just to document what happened. It is there to help shape how the work gets done in the first place.
Rebecca uses Quality to understand which risks need to be addressed early and development approaches that avoid later rework.
As devices become more complex, with closed-loop systems, sensing, algorithms, and AI-driven decision making, how does the role of Quality evolve beyond documentation and into product strategy?
This is where things get really interesting.
Historically, a lot of Quality work in medical devices was focused on design controls, process validation, reproducibility, and making sure hardware was built consistently and safely. That still matters. But once you start bringing in closed-loop systems, algorithms, AI, and more dynamic behavior, the questions Quality needs to address become much broader.
Now you are not just validating a device in the traditional sense — you are thinking about behavior over time, data integrity, model validation, software changes, cybersecurity, and how risk evolves when a system is more adaptive or more complex. Those are not just technical details sitting off to the side. They start affecting product strategy directly.
That means Quality needs to be involved much earlier. It has to be part of the conversations around what data is needed, how performance will be measured, what kind of evidence the company will need later, and how updates will be handled safely. As devices get smarter, Quality professionals need to understand enough of both the technology and the regulatory side to help shape the roadmap, not just support it after the fact.
For startups building first-in-human or early commercial neurotech devices, what are the most common pitfalls you see in Quality systems, and how can founders avoid them early?For startups building first-in-human or early commercial neurotech devices, what are the most common pitfalls you see in Quality systems, and how can founders avoid them early?
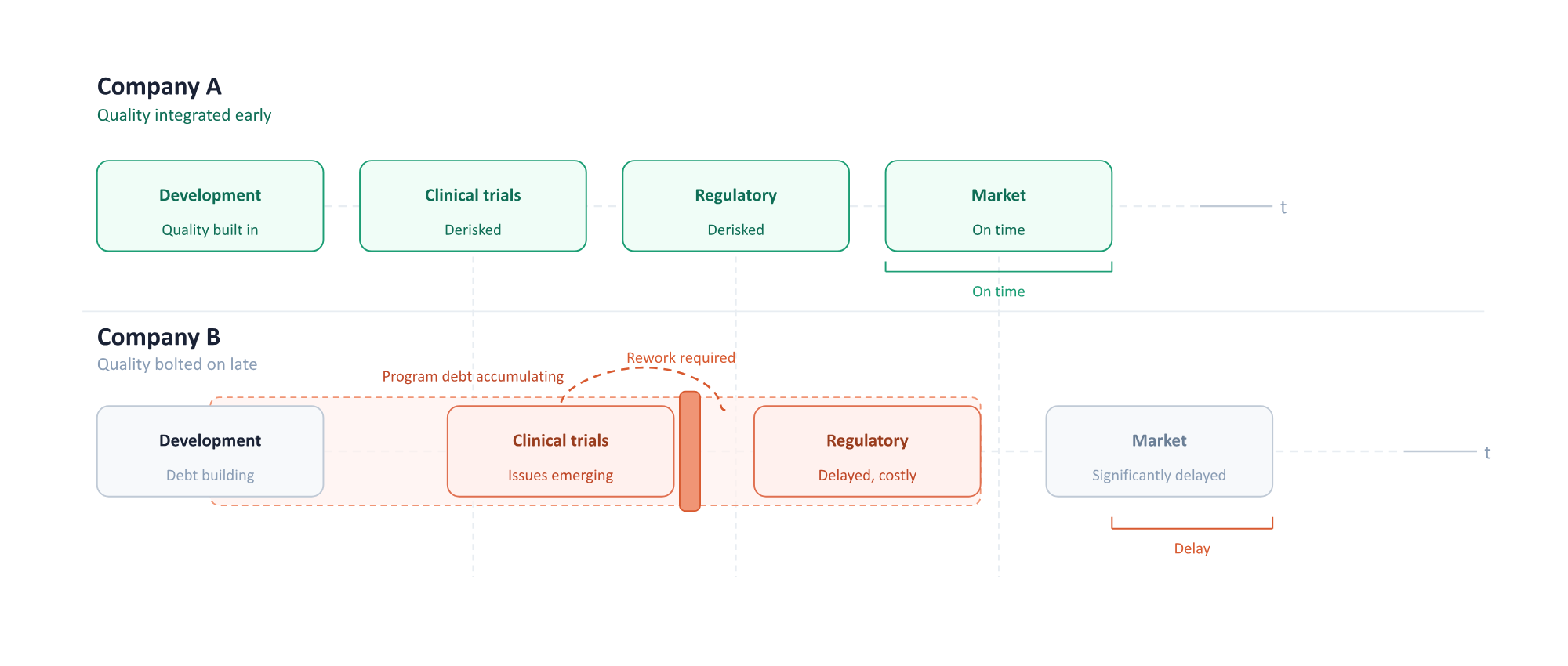
The most common problem is waiting too long.
Founders try to deal with Quality after the product is more defined or clinical trials are closer. But by then, the program debt is expensive and time-consuming or impossible to address without a real schedule hit.
There is a reason Design Controls were introduced, and a reason the FDA expects companies to take them seriously, even during early development. Defining product requirements, documenting decisions, and thinking through risk is not just about compliance. It actually helps the company get clearer on what it is building and why. I have been in plenty of requirements meetings where the process itself revealed the team was not fully aligned on what the product actually needed to do.
Start with something lightweight but scalable. Build a framework early, document decisions as you make them, and tie Quality development into the roadmap instead of treating it as a separate function you can bolt on later. And get experienced help earlier than you think you need it. A small amount of guidance from someone who has seen these patterns before can save a lot of time and struggle later.

Dealing with Quality only when a product is well-defined can lead to program debt that is expensive and time-consuming to address.
How should CEOs and engineering leaders think about Quality as a competitive advantage, not just as a compliance requirement, especially when scaling manufacturing or entering new markets?
Think of it as part of the company’s foundation, not an overhead cost.
Strong Quality systems help companies move faster because they reduce confusion, rework, and avoidable mistakes. When risks are being managed early and processes are under control, regulatory reviews go more cleanly and scale-up has fewer surprises.
It also signals maturity to investors. A stage-appropriate QMS tells investors that the company is building seriously and thinking ahead.
Then once you start scaling, Quality becomes even more important. You need to be able to transfer processes to new suppliers, manage design changes, justify material shifts, and respond quickly when problems come up. None of that works well without a strong Quality foundation.
“ As neurotech keeps evolving, Quality will have to evolve with it. ”
Looking ahead, where do you think neurotech Quality is headed in the next 5 to 10 years as AI, miniaturization, and digital therapeutics mature? What excites you most about that future?
As neurotech keeps evolving, Quality will have to evolve with it. The traditional frameworks still matter, but on their own they are not going to be enough.
AI brings a new set of challenges around validation, transparency, monitoring, and lifecycle management. Miniaturization creates tighter tolerances and more complexity in manufacturing. And with wearables and digital therapeutics, we are also seeing more overlap with software and consumer-tech ways of working, which is going to keep influencing how Quality is approached.
What excites me most is the patient side of all this. Neurotech is moving into areas with so much unmet need, such as stroke recovery, depression, and addiction, where people still don’t have good options. These technologies need to make it through development, which means more teams need to build well from the beginning. Lowering that barrier is a big part of what drives the work we do at Crane Medtech Partners. If we get that right, more good technologies reach the clinic and that is what really matters.
Also published on Medium via NeuroTechX
Carter Sciences delivers personalized talent strategies backed by 20 years of international headhunting experience. We support growth-stage startups with tailored solutions and cost-effective fee structures, so you can scale without impacting your runway.
Specialisms include: Neurotechnology, Neuromodulation/Stimulation, Brain-Computer Interfaces, Wearable Devices, Neurosurgical Technology, and Private Equity/Venture Capital.
Contact Carter Sciences
